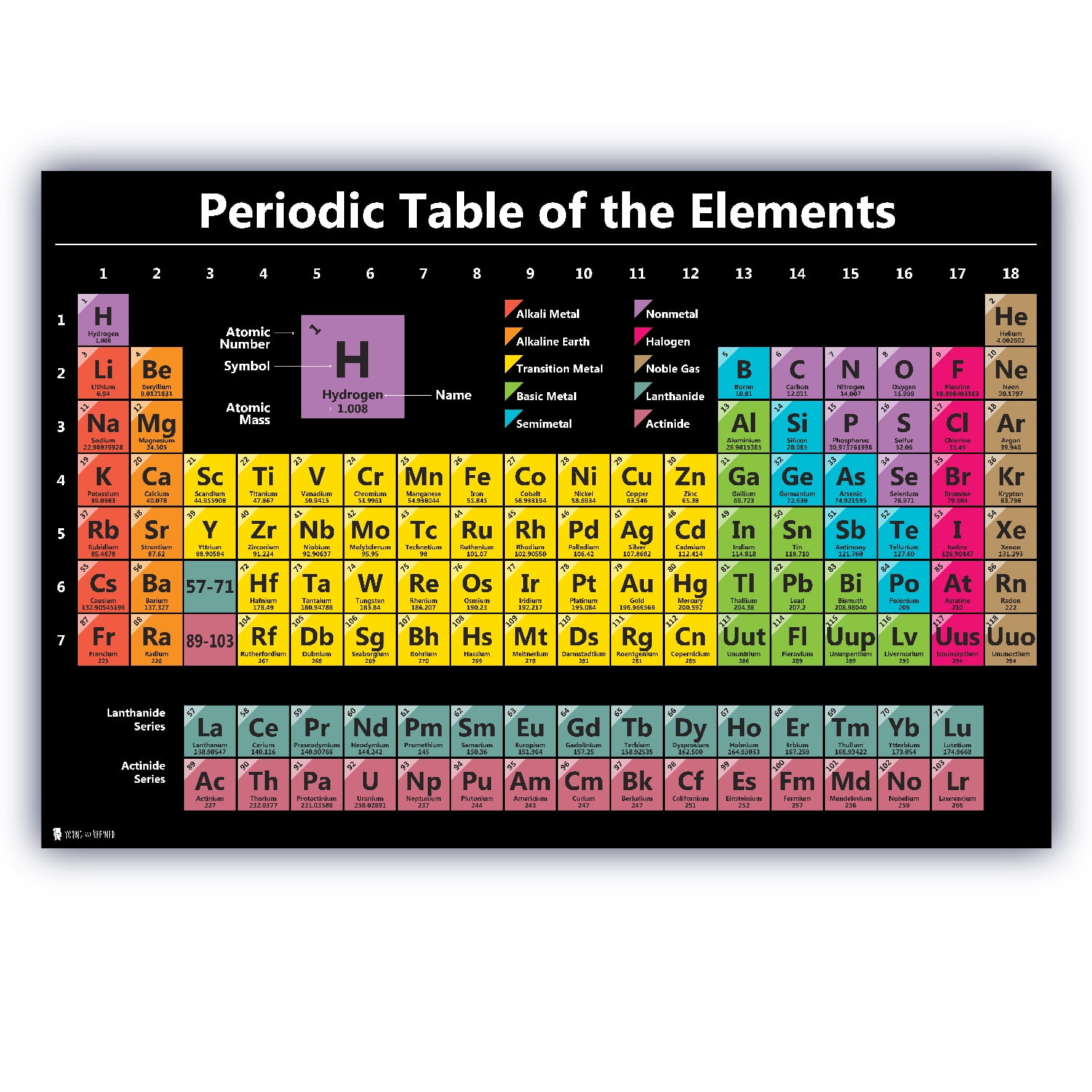
 Development of the periodic table – Use this article to find out about the first scientific discovery of an element in 1649 and how this grew into the periodic table as we know it today.It has the same number of electrons in its outermost energy level as the other elements in Group 1, but as it is a gas, it is normally put in the non-metal group. 
Hydrogen has the atomic number 1, so it sits at the top left of the table with the alkali metals of Group 1. Metals, semi-metals and non-metals – These share some properties, but not enough for a group to fill a column. The atomic numbers of these elements actually sit in the bottom left corner of the main table. Transition metals – Periodic tables have an extra block of transition metals at the bottom, for elements called rare-earths (or lanthanides) and actinides. The groups of elements with similar properties have names and are normally coloured differently in a periodic table. The more you find out about atomic structure, the more patterns and relationships you’ll find in the periodic table. Other patterns are there, too, For example, the energy needed to get an electron away from an atom:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
